2-D scalable ion focus polarization dialyzer
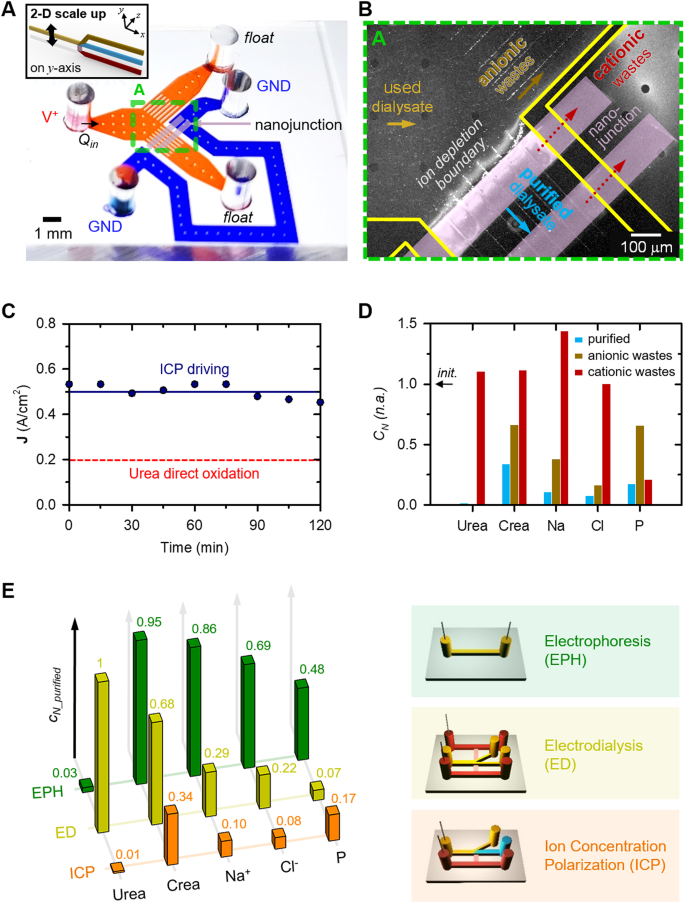
To display scalability of ICP dialyzer in precept, a two-dimensional (2-D) micro-nanofluidic scaling system was fabricated as proven in Fig. 2A. For particulars concerning its supplies used, fabrication course of and experimental setup, discuss with the part of Experimental strategies and Supplementary Data (SI) Fig. 1A. To accommodate the Renal Panel’s requirement of a minimal of 100 µL undiluted pattern for measuring parts in a dialysate resolution, we optimized effectivity by mitigating electrokinetic instability by micro-fin constructions close to the bifurcation level [35] and decreasing electrical resistance utilizing double-patterned nanojunctions. Human used dialysate resolution was repeatedly injected into each anodic and cathodic microchannels at a gradual movement fee of 0.4 µL/min. Underneath these circumstances, ICP phenomenon was generated, resulting in the event of an ion depletion boundary forward of the nanojunction. This separation mechanism utilized for wastes removing and purification, as depicted in Fig. 2B. Notably, a secure present measurement was sustained for 120 min, as demonstrated in SI Fig. 1B. Anionic fluorescent dye molecules (Alexa 488, Invitrogen, USA) and carboxylate micro-particles (1 μm diameter, Invitrogen, USA) have been unable to traverse the ion depletion boundary, redirecting them exterior this boundary, as indicated in SI Video 1.
As well as, Fig. 2C was included to underscore that the proposed ICP dialyzer employed a present density able to direct urea oxidation. It has been reported that, underneath excessive potentials exceeding 1.7 V versus the saturated calomel electrode (SCE), urea undergoes decomposition by a direct oxidation course of on the electrode floor, resulting in the formation of nitrogen and carbon dioxide gasoline. This phenomenon is represented by the next equations [37,38,39]: Eq. (1) outlines the anodic response, Eq. (2) delineates the cathodic response, and Eq. (3) represents the general response.
$${rm{CO}}{left( {{rm{N}}{{rm{H}}_{rm{2}}}} proper)_{rm{2}}}{rm{ + 6O}}{{rm{H}}^{rm{ – }}} to {{rm{N}}_{rm{2}}}{rm{ + 5}}{{rm{H}}_{rm{2}}}{rm{O + C}}{{rm{O}}_{rm{2}}}{rm{ + 6}}{{rm{e}}^{rm{ – }}}$$
(1)
$${rm{6}}{{rm{H}}_{rm{2}}}{rm{O + 6}}{{rm{e}}^{rm{ – }}} to {rm{3}}{{rm{H}}_{rm{2}}}{rm{ + 6O}}{{rm{H}}^{rm{ – }}}$$
(2)
$${rm{CO}}{left( {{rm{N}}{{rm{H}}_{rm{2}}}} proper)_{rm{2}}}{rm{ + }}{{rm{H}}_{rm{2}}}{rm{O}} to {{rm{N}}_{rm{2}}}{rm{ + 3}}{{rm{H}}_{rm{2}}}{rm{ + C}}{{rm{O}}_{rm{2}}}$$
(3)
Right here, the electrochemical decomposition yields nitrogen and carbon dioxide gases, that are thought-about biologically inert with no opposed results on human well being. To supply experimental proof supporting urea direct oxidation utilized by the ICP phenomenon, the gas-to-liquid quantity ratio of N₂ and CO₂ generated from the anodic urea electrochemical decomposition response (1) was first calculated. When 0.29 g/mL of urea was added to contemporary dialysate, the gas-to-liquid ratio was decided to be roughly 14.8–17.6%. The detailed calculation course of was offered in SI Fig. 2. Subsequently, experimental validation was carried out utilizing a tool as illustrated in SI Fig. 2A. From the effluent of the anodic aspect channel, steady gasoline manufacturing was noticed as depicted in SI Fig. 2B. The gas-to-liquid quantity ratio was quantified as roughly 5–8%, primarily attributed to Faradaic losses. These gasoline manufacturing outcomes offered oblique proof supporting urea direct oxidation utilized by the ICP phenomenon. Notice that when urea undergoes decomposition, nitrogen oxidants are shaped, which can pose a danger to human well being underneath particular circumstances: both at potentials beneath 1.6 V versus SCE or underneath pure oxidation circumstances.
Subsequent, every stream was individually extracted, and quantifiable focus profiles of key indicators – urea, creatinine, Na+, Cl–, and phosphorus (P) – vital for affected person well being evaluation, have been established, as proven in Fig. 2D. The focus modifications of every key indicator have been nondimensionalized as follows.
$${C_N} = {matrix{ focus,of,every,indicator hfill cr ,at,the,outlet,({C_{indicator_outlet}}) hfill cr} over matrix{ focus,of,every,indicator hfill cr ,at,the,inlet,({C_{indicator_inlet}}) hfill cr} }$$
First, the positively charged species (Na+ and creatinine) have been faraway from the purified stream relying on their electrophoretic mobility [36]. A considerable proportion of Na+ ions transited the nanojunction by way of cationic flux, yielding a closing assortment of 90% desalted stream (normalized focus of 0.1). Whereas creatinine (sub nanometer molecule and one of many main toxins of physique wastes from a used dialysate) is impartial at pH 7.4, it acquires a optimistic cost underneath pH 7.4. Given the marginally acidic nature of the dialysate, we confirmed that creatinine follows a cationic-like transport mechanism throughout the ICP phenomenon improvement. Creatinine focus decreased each exterior (yellow channel) and inside (blue channel) the ion depletion boundary on the anodic aspect, however elevated on the cathodic aspect (brown channel). Roughly 50% of creatinine crossed the nanojunction, with round 33% passing by the stream exterior the ion depletion boundary. Finally, round 17% of creatinine remained inside the ion depletion boundary, resulting in a big discount within the purified stream focus (normalized focus of 0.34). Second, the negatively charged chloride ions (Cl–) underwent electrochemical reactions on the anodic electrode to take care of electro-neutrality as a result of ICP phenomenon, inflicting a redistribution of focus profiles close to the nanojunction. Third, urea, an uncharged molecule and a significant toxin in physique wastes alongside creatinine, underwent full elimination by electrochemical reactions in anodic aspect streams, together with the purified stream (normalized concentrations of 0.01). Final, the weakly charged phosphorus (P) was considerably faraway from each anodic and cathodic channels (normalized concentrations of 0.17). To know the distinct removing mechanism of P in comparison with different parts, we monitored the motion of the ion depletion boundary throughout a 3-hour system operation, as offered in SI Fig. 1C. The principal constituent of those precipitates was recognized as phosphorus as demonstrated in SI Fig. 1D. Thus, we deduced that P didn’t traverse the ion depletion boundary or the nanojunction; quite, it underwent decomposition resulting from an electrical area, subsequently being expelled by the anionic wastes channel.
As depicted in Fig. 2E, we tried to search out candidates for transportable peritoneal dialyzer utilizing micro-nanofluidic units fabricated primarily based on three totally different mass switch physics. First, we fabricated a easy microfluidic system that was ruled solely to electrochemical reactions and electrophoresis (known as “EPH” in Fig. 2E). On this setup, two electrodes have been positioned inside a single microchannel: one in proximity to the inlet reservoir and the opposite adjoining to the outlet reservoir, with no interconnecting junctions. The microchannel was loaded with used dialysate, and a mix of optimistic voltage and strain was utilized to the inlet reservoir. Underneath these circumstances, full elimination of urea was achieved, whereas creatinine removing reached roughly 5%, Na+ removing roughly 14%, Cl– removing roughly 31%, and P removing roughly 52%. The decisive issue for the whole removing of urea could be attributed to an electrochemical response.
Second, to imitate the ED cell construction, three totally different microchannels and nanojunctions have been constructed throughout them (known as “ED” in Fig. 2E). One microchannel was designated for anodic processes situated on the higher aspect, one other for the managed movement of a pattern alongside the center path, and a 3rd for cathodic processes located on the decrease aspect. Inside this association, it was noticed that the ion depletion efficiently manifested inside the middle-bifurcated channel. This was as a result of institution of a strong electrical area throughout the nanojunction between the higher anodic channel and the decrease cathodic channel. Notably, urea skilled no alteration as a result of absence of electrodes within the center channel, whereas creatinine and Na+ ions have been eliminated by way of electrical transport pushed by the ICP phenomenon.
Lastly, the circumstances of ICP was employed inside a micro-nanofluidic system. As talked about earlier, the whole elimination of urea and the removing of different substances have been achieved by electrical transportation utilized by the ICP phenomenon (known as “ICP” in Fig. 2E). The applying of a decrease potential hindered the removing effectivity of all parts.
Primarily based on the insights gleaned from these analyses, we concluded that the purification of dialysate can solely be achieved when the ICP phenomenon was induced by the presence of an electrode inside the stream of dialysate (known as “ICP”). We named these micro-nanofluidic units as a 2-D scalable ICP dialyzer. As well as, by enhancing the throughput capability from 0.1 µL/min (inset Fig. 1C) to 0.4 µL/min (Fig. 2B), we have been capable of conduct exact analyses by minimizing errors attributable to extended experiments for making certain minimal pattern quantity requirement of the Renal Panel measurement.
(A) A microscopic picture of 2-D scalable ICP dialyzer. Polymeric materials, PDMS was used for constructing microchannels and nanoporous membrane, Nafion was used for patterning nanojunctions, and the microchannel width was expanded utilizing a micro-fin construction which suppressed an electroosmotic instability (EOI). (B) A microscopic picture of dialysate purification as a result of ICP phenomenon. Anionic wastes have been rerouted exterior the ion depletion boundary and cationic wastes have been eliminated by the nanojunction by cationic flux in order that purified dialysate was extracted from stream contained in the ion depletion boundary. (C) A measurement results of present densities versus time when 70 V was utilized for ICP technology. The present densities used for separation of the purified and anionic wastes streams exceeded the identified present densities for urea direct oxidation [38]. (D) Nondimensionalized focus profiles of main dialysis indicators inside (blue) and out of doors (brown) the ion depletion boundary of the anodic aspect streams, and the cathodic aspect stream (pink). Concentrations of all indicators within the used dialysate decreased to beneath 30% within the purified channel (blue bar). (E) Graph exhibiting the outcomes of a management experiment to confirm the dialysate purification mechanism. Dialysate purification was profitable just for ICP situation
Growth of 3-D ion focus polarization dialyzer
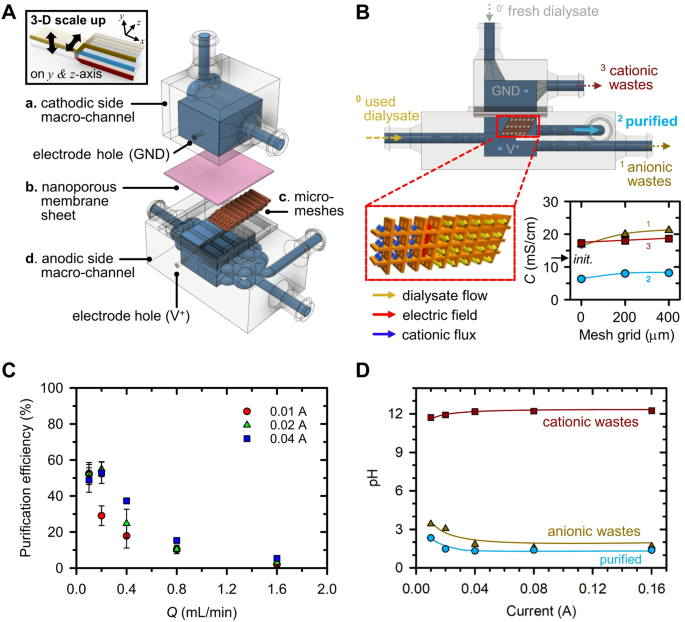
Whereas the feasibility of ICP dialyzer was demonstrated inside a micro-nanofluidic hybrid configuration, the throughput of the system proved insufficient for sensible dialysate recycling, both for human or animal testing functions. On this chapter, we now have engineered a macro-scale ICP dialyzer with a throughput capability of milliliters per minute, as illustrated in Fig. 3A. This system contains: (a) a cathodic aspect macro-channel, that includes an inlet for introducing contemporary dialysate and an outlet for the removing of cationic wastes; (b) a nanoporous membrane sheet, serving to take away positively charged species; (c) a nanoporous resin-coated micro-mesh construction, supposed to mitigate electroosmotic instability (EOI) and increase cationic flux; and (d) an anodic aspect macro-channel, incorporating an inlet for used dialysate and shops for every purified and anionic wastes stream.
The elemental idea revolved round establishing a microfluidic area inside a macro-fluidic equipment [40]. The enlargement of the ICP layer was crucial for efficient dialysate filtration on this high-throughput configuration. Nonetheless, if the channel dimensions exceeded O(100) µm, analogous to different micro-nanofluidic units, the system’s purification efficacy waned [35, 41]. ICP formation within the micro-nanofluidic regime was demarcated by three principal traits: (1) floor conduction (SC), (2) electroosmotic movement (EOF), and (3) electroosmotic instability (EOI), all of which exerted a pivotal affect on ion conveyance throughout the nanojunction [42]. Relying on the channel’s attribute size, SC, EOF, and EOI held sway over ion transportation in very slender channels (< 5 μm), reasonably slender channels ( < ~ O(100) µm), and wider channels ( > ~ O(100) µm), respectively. Consequently, this macroscale system was anticipated to exhibit EOI traits. Nonetheless, it’s essential to notice that EOI-dominated methods inherently endure from avoidable instability [43,44,45] and heightened vitality consumption resulting from decreased overlimiting conductance [46]. To transition the system from an EOI-dominant state to EOF or SC, micro-fin constructions have been beforehand employed in a two-dimensional micro-nanofluidic setup [35]. These fins successfully curbed EOI and allowed for the mixing and elevation of throughput as much as ranges comparable to traditional ICP units. Nonetheless, channel enlargement solely in a single airplane was inadequate to attain greater than laboratory-scale throughput.
To beat this limitation, we prolonged the fins within the z-direction (i.e., three-dimensionally), leading to micro-meshes as depicted in Fig. 3B. This mesh was meticulously devised to generate an electrical area perpendicular to the dialysate movement path, thereby enabling cationic wastes transport alongside the electrical area’s orientation. Moreover, an optimized micro-mesh grid dimension was decided by experimentation with three variants: (1) no micro-mesh, (2) 200 μm, and (3) 400 μm micro-meshes, as detailed in SI Fig. 3A. Optimum purification effectivity was realized with a micro-mesh possessing a 400 μm grid dimension, as this configuration exhibited probably the most pronounced distinction in conductance between purified and anionic wastes streams. To develop the cation transport area inside the macroscale system, we utilized a nanoporous resin coating to the micro-mesh construction. Microscopic photographs of the micro-meshes earlier than and after the nanoporous resin coating have been offered in SI Fig. 3B, confirming profitable coating inside the open areas. The conductance outcomes, displayed in SI Fig. 3C demonstrated the affect of the nanoporous resin coating on the micro-mesh construction. When the nanoporous resin was coated on the micro-meshes, the conductance of anionic wastes and cationic wastes streams elevated, indicating enhanced cationic flux.
Subsequently, we discovered the optimum present and movement fee circumstances that yielded the very best dialysate purification effectivity for the preliminary model of the 3-D scaling-up system, as depicted in Fig. 3B. The precise picture of the Fig. 3B was offered in SI Fig. 3D. As a result of traits of the dialyzer, fluid-cell interplay happens, making it essential to make use of biocompatible supplies [47]. Subsequently, a 3-D ICP dialyzer was fabricated utilizing biocompatible materials appropriate for medical functions. Supplies used, fabrication and experimental particulars have been written within the part of Experimental strategies, respectively. First, as proven in SI Fig. 4A, the conductance of the purified, anionic wastes, and cationic wastes streams was measured in keeping with modifications in present and movement fee. With the intention to comprehensively analyze the rise in conductance as a result of enhance of anionic wastes within the anionic wastes stream and the lower in conductance as a result of lower of each anionic and cationic wastes within the purified stream, purification effectivity was outlined as follows and plotted in Fig. 3C.
$$eqalign{& Purification,effectivity cr & = left( {1 – {matrix{ conductance,of hfill cr purified,stream hfill cr} over matrix{ conductance,of hfill cr ,anodic,wastes,stream hfill cr} }} proper) occasions 100(% ) cr} $$
Underneath the I = 0.01 A utility, the purification effectivity decreased exponentially because the movement fee elevated, and underneath the I = 0.02 A and I = 0.04 A utility, the very best purification effectivity was obtained at Q = 0.2 mL/min and was comparatively decrease underneath the I = 0.04 A utility.
Second, as proven in Fig. 3D, we confirmed the pH modifications because the utilized present elevated underneath the situation of Q = 0.2 mL/min, which confirmed maximal purification effectivity. Right here, we used Pt and Ag as anode and cathode electrodes materials, respectively, primarily based on the ends in SI Fig. 4B. Because the utilized present elevated, the lower in pH of the anionic wastes and purified streams elevated, and above I = 0.04 A, they converged and maintained an analogous pH worth. In the meantime, the pH of the cationic wastes stream elevated with the rise in utilized present, and converged above I = 0.04 A. Primarily based on the outcomes of Fig. 3 C and D, the optimum working circumstances of the three-dimensional (3-D) ICP dialyzer have been established as I = 0.02 A, Q = 0.2 mL/min, the place the pH of the purified stream was comparatively excessive and the purification effectivity was the utmost. This electrical and mechanical situation ensured a harmonious steadiness between excessive movement fee and secure purification effectivity.
(A) Schematic diagram of 3-D scalable ICP dialyzer. The system consisted of a cathodic aspect macro-channel with inlet for contemporary dialysate and outlet for cationic wastes flowing, a nanoporous membrane sheet for eradicating positively charged species, micro-meshes for decreasing EOI and enhancing cationic flux an anodic aspect macro-channel with inlet for used dialysate and shops for purified and anionic wastes flowing. (B) Schematic diagram and graph of micro-mesh construction design optimization. Coated nanoporous resin on the micro-meshes floor ensured that the cationic wastes have been transported in direction of the nanoporous membrane sheet alongside the path of the electrical area in order that purified dialysate handed by the micro-meshes perpendicular to the electrical area. The micro-mesh grid dimension of 400 μm was chosen for the reason that distinction in conductance between purified and anionic wastes streams was the best, and conductance of cationic wastes was the very best. (C) Graph exhibiting modifications in purification effectivity in keeping with modifications in present and movement fee. Most purification effectivity was noticed when making use of I = 0.02 A and Q = 0.2 mL/min. (D) Graph exhibiting modifications in pH because the utilized present elevated underneath the situation of Q = 0.2 mL/min. Because the utilized present worth elevated, the pH of anionic wastes and purified streams decreased, and that of cationic wastes elevated
3-D scalable ion focus polarization dialyzer
The beforehand fabricated 3-D ICP dialyzer, which had a movement fee of Q = 0.2 mL/min, was additional scaled as much as obtain a Q = 1 mL/min, as proven in Fig. 4A, and an precise picture of the system was proven in SI Fig. 3E. The fabrication course of and meeting elements of the system have been the identical, solely the elements dimension was expanded within the y-axis path. As illustrated in Fig. 4B, the design parameters of the system have been scaled proportionally in relation to the throughput enhance (0.2, 0.4, and 1 mL/min), together with the full quantity inside the system (Vwhole), the amount excluding the mesh body (Vwhole−mesh), and the nanoporous membrane’s contact space (Acontacted membrane) with the fluid. Primarily based on acceptable proportional constants, a tool with a most throughput of 1.0 mL/min was realized. Additional evaluation concerned the facility consumption of scaled units, as proven in Fig. 4C. Though greater throughput correlated with decrease energy consumption, the variations have been modest (inside 10%), and the temporal tendencies remained constant. Accumulation of gasoline byproducts across the anodic electrode heightened electrical resistance, with the 1 mL/min system demonstrating the bottom energy consumption as a result of ample outlet area for gasoline dispersal. Over time, the presence of trapped gases close to the electrode led to rising energy consumption, finally reaching a saturation level. Regardless of parallel design and manufacturing of units, discrepancies in dialysis efficiency may come up.
Determine 4D confirmed the conductance of streams from units with various throughput capacities following the infusion of contemporary dialysate. Within the case of purified stream, the conductance exhibited a constant lower of round 10% in comparison with the launched contemporary dialysate, plateauing at roughly 10 mS/cm whatever the movement fee (0.2, 0.4, 1 mL/min). Conversely, the conductivity of the anionic wastes stream decreased with rising remedy capability. This development was attributed to lowered electrode-fluid contact space per unit capability, leading to decreased electrochemical response and wastes removing effectivity [48]. For cationic wastes stream, the conductance remained secure at roughly 15 mS/cm, regardless of throughput capability. This consistency was ascribed to the nanoporous membrane’s capacity to take care of a uniform ion transport fee per unit space from the anodic to the cathodic channel.
Subsequent evaluation concerned the efficiency of ICP dialysis utilizing human peritoneal dialysate, as depicted in Fig. 4E. Removing ratios for urea, creatinine, Na+, Cl–, and P have been measured from purified stream. Whereas urea’s removing ratio exhibited heightened variability with rising throughput capability, it maintained a mean of over 50%. In distinction, the typical removing ratio for anionic wastes stream was 99% (SI Fig. 5A), indicating localized decomposition of urea close to the electrode. Removing ratio of Na+ remained constant at round 40%, whereas creatinine, characterised as a weak natural cation, confirmed a removing ratio beneath 25%, which elevated with greater throughput capacities. Cl– exhibited common removing ratios of 20% for the 0.2 mL/min system and 25% for the 0.4 and 1 mL/min units. P removing ratios demonstrated an rising development with greater throughput capacities. Focus modifications within the cationic wastes channel have been offered in SI Fig. 5B, highlighting elevated concentrations of Na+ and creatinine resulting from their cationic nature upon passing by the nanoporous membrane sheet.
Whereas Fig. 4E confirmed the fast measurement of collected resolution upon powering on, Fig. 4F offered measurements taken from 10 to 60 min thereafter utilizing the 3-D ICP dialyzer with Q = 1 mL/min. Noticeably, the removing ratio of all indicators exhibited constant upkeep over time, barring creatinine, and urea removing ratio was dropped beneath 20%. On this examine, we used electrode supplies with the identical size and floor space whatever the system dimension. Urea was decomposed on the electrodes, and whereas bubble formation on the electrode floor was not outstanding within the 0.2 mL/min system, it observably elevated within the 1 mL/min system. The bubbles amassed across the electrodes lowered the efficient electrode floor space required for urea decomposition and hindered electrical paths for creatinine transportation by the nanoporous membrane. As indicated in SI Fig. 5A, the urea removing ratio remained near 99% even within the 1 mL/min system for the anionic wastes stream, collected close to the electrode, suggesting that the problem lies not with electrode efficiency however with system optimization. Primarily based on these experimental evidences, we inferred that urea decomposition may very well be stably achieved by (1) putting in a bubble removing membrane across the electrode, (2) designing the hole between the electrode and purified channel extra intently, or (3) rising the electrode floor space.
ICP Dialyzer-assisted peritoneal Dialysis
Lastly, to check the in vivo toxicity discount efficiency of typical PD and ICP dialyzer-assisted PD, we employed a bilateral nephrectomy rat mannequin. The peritoneal cavity of the rat was too small to repeatedly drain the injected dialysate, so, we carried out quasi-continuous PD to confirm the advance in in vivo toxicity discount when the ICP dialyzer-assisted typical PD, as depicted in Fig. 5A. Submit-surgery, all rats have been allowed a 24-hour recuperation interval, adopted by 2 h of respective PD periods, throughout which concentrations of uremic and different pivotal parts have been monitored. Just for group 3 (rat with bilateral nephrectomy), after injection of contemporary dialysate (0 h), 12 mL of the used dialysate was extracted and infused into the ICP dialyzer for dialysate regeneration. 4 mL of the regenerated purified dialysate was discharged and re-injected into the rat’s peritoneal cavity at 30-minute intervals.
Subsequent, modifications in serum concentrations of all main indicators have been monitored and plotted, as proven in Fig. 5B for group 1, Fig. 5C for group 2, and Fig. 5D for group 3. Serum concentrations have been normalized as follows.
$${C_{N_{serum}}}left( t proper) = {{{C_{serum}}left( t proper)} over {{C_{serum}}left( { – 24 h} proper)}}$$
Throughout the preliminary 24-hour interval, rats with surgically eliminated kidneys (teams 2 and three) exhibited a noticeable escalation in toxin concentrations, in distinction to the negligible change noticed in regular rats (group 1). In teams 2 and three, the concentrations of urea and creatinine elevated sharply throughout the first 24 h after surgical procedure. Following the initiation of typical PD, the speed of enhance in urea and creatinine concentrations decreased in each teams 2 and three, whereas there have been no important modifications in any of the indications in group 1. To evaluate the speed of change in urea and creatinine concentrations in teams 2 and three, the focus derivatives over time have been plotted in Fig. 5E. When PD was assisted by the ICP dialyzer, a definite discount in in vivo toxin concentrations inside the first hour was noticed, whereas no such discount was achieved with out the help. These outcomes instructed that the ICP dialyzer may regenerate dialysate, efficiently aiding PD whereas utilizing a smaller dialysate quantity.
Nonetheless, by the second hour, a rise in in vivo toxin concentrations was noticed in each teams 2 and three. To determine the trigger, we examined the removing ratios over time as proven in Fig. 5F. The outcomes confirmed that the typical removing ratios of urea and creatinine decreased progressively to 31.8%, 13.7%, and a pair of.6% for the primary, second, and third infusion occasions, respectively. Because the used dialysate discharged from the rat’s peritoneal cavity was intermixed with varied seen suspended substances, it amassed contained in the ICP dialyzer, inflicting non-uniformity in movement fee, electrode reactions, and electrical area formation. This interplay lowered the efficient floor space of the nanoporous membrane, resulting in a lower in purification effectivity over time. This impact was notably pronounced for urea and creatinine, that are comparatively bigger in dimension in comparison with electrolyte ions.